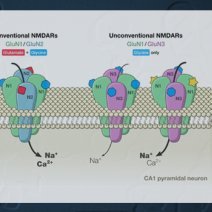
Unmasking GluN1/GluN3A excitatory glycine NMDA receptors.
Teddy Grand, Sarah Abi Gerges, Mélissa David, Marco A. Diana, Pierre Paoletti.
Abstract
GluN3A and GluN3B are glycine-binding subunits belonging to the NMDA receptor (NMDAR) family that can assemble with the GluN1 subunit to form unconventional receptors activated by glycine alone. Functional characterization of GluN1/GluN3 NMDARs has been difficult. Here, we uncover two modalities that have transformative properties on GluN1/GluN3A receptors. First, we identify a compound, CGP-78608, which greatly enhances GluN1/GluN3A responses, converting small and rapidly desensitizing currents into large and stable responses. Second, we show that an endogenous GluN3A disulfide bond endows GluN1/GluN3A receptors with distinct redox modulation, profoundly affecting agonist sensitivity and gating kinetics. Under reducing conditions, ambient glycine is sufficient to generate tonic receptor activation. Finally, using CGP-78608 on P8-P12 mouse hippocampal slices, we demonstrate that excitatory glycine GluN1/GluN3A NMDARs are functionally expressed in native neurons, at least in the juvenile brain. Our work opens new perspectives on the exploration of excitatory glycine receptors in brain function and development.
Nat Commun. 2018 Nov 13 ;9(1):4769. doi : 10.1038/s41467-018-07236-4.