Quantitative Nanoscopy of Inhibitory Synapses: Counting Gephyrin Molecules and Receptor Binding Sites
Christian G. Specht, Ignacio Izeddin, Pamela C. Rodriguez, Mohamed El Beheiry, Philippe Rostaing, Xavier Darzacq, Maxime Dahan and Antoine Triller
The strength of synaptic transmission is controlled by the number and activity of neurotransmitter receptors. However, little is known about absolute numbers and densities of receptor and scaffold proteins and the stoichiometry of molecular interactions
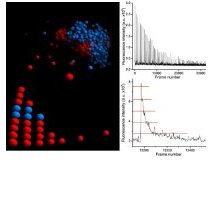
at synapses. Here, we conducted three-dimensional and quantitative nanoscopic imaging based on single-molecule detections to characterize the ultra-structure of inhibitory synapses and to count scaffold proteins and receptor binding sites. We observed a close correspondence between the spatial organization of gephyrin scaffolds and glycine receptors at spinal cord synapses. Endogenous gephyrin was clustered at densities of 5,000–10,000 molecules/mm2. The stoichiometry between gephyrin molecules and receptor binding sites was approximately 1:1, consistent with a two-dimensional scaffold in which all gephyrin molecules can contribute to receptor binding. The competition of
glycine and GABAA receptor complexes for synaptic binding sites highlights the potential of single-molecule imaging to quantify synaptic plasticity on the nanoscopic scale.
Neuron. 2013 Jul 24;79(2):308-21