Targeting and imaging single biomolecules in living cells by complementation-activated light microscopy with split-fluorescent proteins
Fabien Pinaud and Maxime Dahan
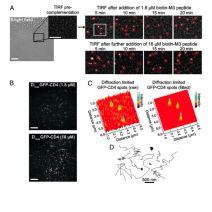
Single-molecule (SM) microscopy allows outstanding insight into biomolecular mechanisms in cells. However, selective detection of single biomolecules in their native environment remains particu- larly challenging. Here, we introduce an easy methodology that combines specific targeting and nanometer accuracy imaging of individual biomolecules in living cells. In this method, named com- plementation-activated light microscopy (CALM), proteins are fused to dark split-fluorescent proteins (split-FPs), which are acti- vated into bright FPs by complementation with synthetic peptides. Using CALM, the diffusion dynamics of a controlled subset of ex- tracellular and intracellular proteins are imaged with nanometer precision, and SM tracking can additionally be performed with fluorophores and quantum dots. In cells, site-specific labeling of these probes is verified by coincidence SM detection with the complemented split-FP fusion proteins or intramolecular single- pair Förster resonance energy transfer. CALM is simple and com- bines advantages from genetically encoded and synthetic fluores- cent probes to allow high-accuracy imaging of single biomolecules in living cells, independently of their expression level and at very high probe concentrations.
Proc Natl Acad Sci U S A. 2011 Jun 14;108(24):E201-10.